Lately, it seems that “CO2 extracts” are being mentioned on almost every aromatherapy page. What are CO2 extracts? How are they made? Do we even consider them essential oils? To better understand the CO2 extraction process, let’s review some other ways that essential oils are obtained.
Steam Distillation
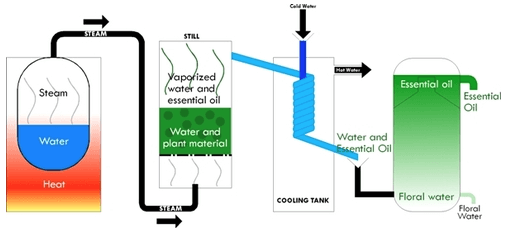
We obtained most of the essential oils we know and love by way of steam distillation, done inside of a “still.” The basic process involves four separate parts. First, we bring heated water to a boil in the first chamber. The steam that’s produced becomes forced into a second chamber containing plant material, such as lavender flowers or peppermint leaves. An essential oil that is found inside plant cells is forced out by the heated steam. Then, the essential oil vaporizes, joins with the steam, and carries across a cooling mechanism called the condenser.
Next, the condenser cools the mixture, which results in a change from a gaseous form (steam) to a liquid state (water and liquid oil). When the vapors condense back into the liquid state, they drip into a third chamber called the condensation or collection chamber. Since we know that oil and water don’t mix, you can guess what happens next. The essential oil layer floats on top of the water layer (hydrosol, also called hydrolat). After separating the two layers, the essential oil is ready to be bottled[1].
Cold-Pressing
Citrus fruits contain essential oil in the glands of the outer peel. Cold-pressed essential oils are mechanically punctured through either the citrus peels or the whole fruit and collect the expressed liquid. The oil and aqueous (water/juice) layers then separates[2].

Solvent Extraction
Solvent extraction for absolutes is technically not considered essential oils. Extracted via a multi-step process that utilizes more delicate plant materials (such as jasmine flowers) that wouldn’t survive the heat of steam distillation.
Like absolutes, CO2 extracts aren’t considered true essential oils, such as those obtained by steam distillation and cold-pressing. Carbon dioxide (chemical abbreviation= “CO2”) is a gas familiar to most people: it’s what we exhale during breathing. We breathe oxygen into the lungs during inhalation and exhale carbon dioxide. Carbon dioxide is a waste product of the body’s metabolism. But how does carbon dioxide help produce oil?
Under pressure, CO2 gas transforms into an “almost” liquid state called the supercritical state. Plant material goes through an airtight receptacle, and carbon dioxide gas is pumped under pressure; low heat is also applied to aid extraction. As the pressure inside the container rises, the CO2 gas nearly liquifies, bathing the plant material in supercritical CO2. The combination of high pressure and low temperatures encourages the plant material to releases its aromatic components. After a period of time, the pressure reduces and the supercritical CO2 then changes back to its gaseous state, completely dissipating from the extracted material.
Because lower temperatures occur during the process, heat-sensitive plant components may destroy or inactivated during steam distillation are preserved in the CO2 extract. The combination of lower temperatures and (generally) lower pressure means that CO2 extracts contain more of a plant’s original constituents than steam distilled oils. According to Dutch aromatherapist Madeleine Kerkhof-Knapp Hayes, the scent of CO2 extracted oils is [generally] “richer and more intense because more aromatic components are present”[5].

Plant Therapy’s CO2 Extracts
A perfect example of this is a wonderfully aromatic Ginger Root CO2 extract carried by Plant Therapy. It does not contain the components gingerol or shogaol, which in part give freshly peeled ginger root its characteristic scent. These components are present in CO2 extracted ginger, which gives it a more pleasing scent than the steam distilled oil. CO2 extracted ginger is also correspondingly “hotter” than steam distilled ginger, so extra caution in diluting it is warranted. Plant Therapy also carries a lovely Vanilla CO2 and Turmeric CO2 extract.
CO2 extracts are sub-divided into two categories, “select” and “total.” Select CO2s extract under lower pressure and resemble essential oils in that they are usually fully liquid. This makes them easier to work with when pipetting or diluting. Total CO2 extracts under higher pressure and contain more of the plant’s original components, including waxes, fatty oils, and color pigments.
Total CO2 extractions are more challenging to work with as they are thicker and require special consideration when diluting. Plant Therapy’s CO2 products are of a select variety.
We want you to learn as much as you want to about essential oils and how to use them safely. If you have any questions, comments or other concerns, you’re welcome to email us at Aromatherapist@plantherapy.com. Or come join us on Facebook at Safe Essential Oil Recipes!
References:
[1] Battaglia, S. 2003. The Complete Guide to Aromatherapy. The International Centre of Holistic Aromatherapy, publishers, Brisbane.
[2] Wilson, R. 2002. Aromatherapy-Essential Oils for Vibrant Health and Beauty. Avery publishers, New York.
[3] Pappas, R. 2015. Important Definitions Concerning Aromatic Products. Notes from Essential Oil University Facebook page.
[4] Smith, C. 2014. Essential Oil Extraction 101. www.essentialoilblogging.com.
[5] Kerkhof-Knapp Hayes, M. 2015. Complementary Nursing in End of Life Care. Kicozo publishers, the Netherlands.



